TL;DR
- The Fire Triangle has three elements — heat, fuel, and oxygen: Remove any one of these three, and fire cannot exist. Every fire prevention and suppression strategy traces back to breaking this triangle.
- Fires spread through four mechanisms — conduction, convection, radiation, and direct flame contact: Understanding how fire travels is just as critical as understanding how it starts.
- Most workplace fires begin with ignition sources nobody bothered to control: Hot work sparks landing on oily rags, overloaded electrical panels, improperly stored flammable liquids — the causes are predictable and preventable.
- The Fire Triangle is not just theory — it is your primary fire prevention tool: Every control measure you implement on site is designed to attack one leg of the triangle.
- Extinguishment works by removing one element: Smothering removes oxygen, cooling removes heat, fuel isolation removes the combustible material. Choosing the wrong method costs time and lives.
I was conducting a routine hot work audit inside a tank farm at a refinery on the Gulf coast when I smelled something sharp — hot slag from a grinding operation two levels above had dropped through an open grating and landed on a pile of oil-soaked absorbent pads stacked against a pipe rack. By the time I reached the pile, there was already a small flame licking up the side. The fire watch assigned to the job was stationed at the grinder — not below the grating where the sparks were falling. We hit it with a dry chemical extinguisher in seconds. But I stood there afterward thinking about how close we came to a tank farm fire because three things lined up at the same moment: a heat source nobody tracked, a fuel source nobody removed, and all the oxygen in the open air.
That moment is the Fire Triangle in its simplest and most dangerous form. The Fire Triangle is the foundational model that every fire prevention program, every hot work permit, every housekeeping inspection, and every emergency response plan is built on. If you work in any industry where ignition sources, combustible materials, or flammable atmospheres exist — and that covers nearly every workplace on earth — then understanding these three elements and how fire spreads is not academic. It is the single most practical piece of fire science you will ever use. This article breaks down each element of the Fire Triangle, explains how fires actually spread in real operational environments, and gives you field-tested strategies to break the triangle before ignition ever occurs.

What Is the Fire Triangle? The Fundamental Model of Combustion
The Fire Triangle is the simplest and most universally applied model in fire science. It states that fire — or combustion — requires the simultaneous presence of three elements: heat, fuel, and oxygen. Remove any single element, and combustion cannot occur. Remove any single element from an existing fire, and the fire goes out.
This model has been the backbone of fire safety training across every industry I have worked in — from petrochemical complexes to construction projects to mining operations. The reason it endures is that it works. Every fire prevention control, every suppression system, every emergency procedure you will ever encounter attacks one or more legs of this triangle.
OSHA’s Fire Safety Standard (29 CFR 1910.155–165) and NFPA 10 (Standard for Portable Fire Extinguishers) both ground their requirements in this principle: identify and control the elements of combustion before they converge.
A few clarifications matter for field application. The Fire Triangle describes ordinary combustion — Class A, B, and C fires. For self-sustaining chemical reactions like metal fires (Class D) or fires involving energized electrical equipment, the model still applies but with added complexity. There is also an expanded model called the Fire Tetrahedron, which adds a fourth element — the chemical chain reaction — to explain sustained combustion at a molecular level. But for practical site safety, hazard identification, hot work control, and fire prevention planning, the three-element triangle is your working tool.
Pro Tip: When I brief crews before hot work, I draw a triangle on the whiteboard and ask three questions: “Where is the heat source? Where is the fuel? Where is the oxygen?” If anyone cannot answer all three with specifics, the permit does not get signed. That five-minute exercise has caught more uncontrolled hazards than any checklist I have ever used.
Element 1 — Heat: The Ignition Energy That Starts Combustion
Heat is the energy component of the Fire Triangle. Without sufficient heat energy to raise a fuel to its ignition temperature, combustion cannot begin. But here is what most training programs skip: heat is not just about open flames. In real workplaces, heat sources are everywhere — and the most dangerous ones are the ones nobody thinks of as “heat.”
Common Workplace Heat Sources
Every fire investigation I have been part of starts with identifying the heat source. The list below covers the ignition sources I encounter most frequently across heavy industry, construction, and manufacturing environments:
- Hot work operations: Welding arcs, cutting torches, grinding sparks, brazing flames. Grinding sparks can travel 10 meters horizontally and retain enough energy to ignite flammable vapors or combustible solids on landing.
- Electrical faults: Overloaded circuits, loose connections, damaged insulation, short circuits, and arc flash events. Electrical ignition is the leading cause of warehouse and facility fires globally.
- Friction and mechanical heat: Seized bearings, misaligned conveyor belts, dry-running pumps, overheating motors. These are slow-build heat sources — they smolder before they ignite, giving off warning signs most workers never notice.
- Static electricity: Generated by fluid flow through pipes, powder transfer, belt-driven equipment, and even personnel walking on non-conductive flooring. Static discharge can ignite flammable atmospheres with vapor concentrations within the explosive range.
- Radiant heat and solar gain: Direct sunlight on dark-colored storage containers can raise internal temperatures above the flash point of stored materials. I have documented surface temperatures exceeding 70°C on black steel drums left in open desert storage.
- Chemical reactions: Exothermic reactions, self-heating materials (oily rags, certain agricultural products, coal dust), spontaneous combustion in improperly stored oxidizing chemicals.
- Smoking materials: Still a significant ignition source despite decades of designated smoking area policies. Discarded cigarettes in unauthorized areas remain a recurring fire cause in construction and general industry.
Key Heat Concepts for Field Application
Understanding a few specific heat-related terms will make you sharper at identifying fire risk during inspections and permit reviews:
- Flash point: The lowest temperature at which a liquid produces enough vapor to form an ignitable mixture near its surface. Gasoline has a flash point of approximately -43°C — it is always producing ignitable vapors at normal ambient temperatures.
- Auto-ignition temperature: The temperature at which a material ignites spontaneously without any external spark or flame. Diesel fuel auto-ignites at roughly 210°C — which is why hot exhaust systems near diesel spills are a serious risk.
- Ignition energy: The minimum energy needed to ignite a flammable mixture. Hydrogen requires only 0.017 millijoules — a static spark too small to feel can ignite it.
Pro Tip: During hot work permit inspections, I always check the area below, behind, and on the other side of walls — not just the immediate work zone. Heat conducts through steel. I have seen fires start on the opposite side of a bulkhead from a welding operation because nobody checked what was on the other side of the plate being welded.

Element 2 — Fuel: The Combustible Material That Burns
Fuel is any material that can undergo combustion when exposed to sufficient heat in the presence of oxygen. On a job site, fuels are everywhere. The challenge is not finding them — it is recognizing them for what they are.
The Three States of Fuel
Fuel exists in three physical states, and each behaves differently in a fire scenario. Recognizing the state of the fuel present in your work area directly determines which fire prevention and suppression strategies apply:
- Solid fuels: Wood, paper, cardboard, textiles, plastics, rubber, dust accumulations, insulation materials. Solid fuels must be heated to the point where they release combustible gases (pyrolysis) before they can ignite. This is why solid fuel fires typically start slowly and accelerate.
- Liquid fuels: Gasoline, diesel, solvents, paints, thinners, hydraulic oils, cooking oils. Liquid fuels do not burn as liquids — they burn as vapors released from the liquid surface. The flash point determines how easily that vapor forms. Low flash point liquids are the most dangerous because they are always producing ignitable vapors.
- Gaseous fuels: Methane, propane, acetylene, hydrogen, natural gas. Gaseous fuels mix directly with air and can form explosive atmospheres within their flammable range. They ignite faster and more violently than solids or liquids because no phase change is needed — the fuel is already in its combustible state.
Fuel Properties That Affect Fire Behavior
Not all fuels burn the same way. Two critical properties determine how quickly a fuel contributes to fire growth — and both are things you can assess during a walkdown:
- Surface area to mass ratio: Fine materials ignite faster than bulk materials. A solid oak beam is difficult to ignite with a match. Sawdust from the same oak will ignite in seconds. Steel wool burns. Steel plate does not. Dust explosions occur precisely because airborne particulates have enormous surface area relative to mass.
- Vapor density: Flammable vapors heavier than air (vapor density greater than 1.0) settle into low points — trenches, pits, basements, sumps. Gasoline vapor is roughly 3–4 times heavier than air. Propane is about 1.5 times heavier. This is why gas detectors in facilities are installed at low elevations and why confined space atmospheric testing must include testing at the bottom of the space.
| Fuel Type | Example | Flash Point | Key Risk Factor |
|---|---|---|---|
| Low flash point liquid | Gasoline | -43°C | Always producing ignitable vapor |
| Medium flash point liquid | Diesel | 52°C | Safe at room temp; dangerous when heated |
| Flammable gas | Propane | N/A (gas) | Heavier than air; pools in low areas |
| Combustible solid | Wood | N/A (solid) | Requires sustained heat; large fuel load |
| Combustible dust | Grain dust | N/A (airborne) | Explosive when suspended in air |
I once walked a new warehouse before handover and found 14 separate fuel sources within 30 meters of the electrical distribution room: cardboard packing material stacked against the wall, polyethylene wrap draped over cable trays, a forgotten can of contact adhesive on a shelf, and cleaning rags stuffed behind a panel. None of these were on anyone’s fire risk assessment. The fuel leg of the triangle is the one people forget fastest because combustible materials look like everyday objects — until they are on fire.
Pro Tip: Train your eye to spot “fuel creep” — the gradual accumulation of combustible materials in areas that were clean when the fire risk assessment was done. Housekeeping is not cosmetic. It is fire prevention. Every hot work permit should include a fresh fuel survey within the work radius, not a reference to last month’s inspection.

Element 3 — Oxygen: The Oxidizer That Sustains Combustion
Oxygen is the third and final leg of the Fire Triangle. Normal atmospheric air contains approximately 21% oxygen — more than enough to support combustion of virtually all common fuels. Most workplace fires burn in open air, which means the oxygen leg of the triangle is almost always present by default. That is why most fire prevention strategies focus on controlling heat and fuel rather than oxygen.
However, there are critical scenarios where oxygen concentration is the deciding factor between a safe environment and a catastrophic one:
- Oxygen-enriched atmospheres: Atmospheres above 23.5% oxygen dramatically accelerate fire behavior. Materials that are difficult to ignite in normal air — Nomex coveralls, leather gloves, even steel — can burn vigorously in oxygen-enriched conditions. Oxygen enrichment occurs around leaking oxygen lines, in medical facilities, near oxygen cutting and welding setups, and in poorly ventilated spaces where oxygen is being used.
- Oxidizing chemicals: Substances like hydrogen peroxide, potassium permanganate, ammonium nitrate, and concentrated nitric acid supply their own oxygen during decomposition. Fires involving these materials cannot be extinguished simply by smothering — the fuel is generating its own oxidizer.
- Oxygen-deficient atmospheres and inerting: Reducing oxygen below approximately 16% will extinguish most hydrocarbon fires. This principle is used in fire suppression systems (nitrogen flooding, CO₂ systems) and in inerting tanks and vessels before hot work. But oxygen levels below 19.5% are immediately dangerous to human life — so oxygen displacement as a fire control creates a confined space atmospheric hazard.
NFPA 53 (Recommended Practice on Materials, Equipment, and Systems Used in Oxygen-Enriched Atmospheres) provides detailed guidance on managing ignition risks in oxygen-enriched environments — an essential reference for anyone working with oxygen systems in industrial, medical, or laboratory settings.
During a turnaround at a gas processing facility, I observed a crew preparing for hot work on a line that had been nitrogen-purged. The atmospheric test showed 3% oxygen — well below the level needed for combustion. The space was safe from fire. But two crew members were about to enter without supplied-air respirators because “the gas test was clear.” The oxygen leg of the triangle had been broken for fire safety — but it had also been broken for human survival. That is the tension field practitioners must manage every day.
Pro Tip: Never assume ambient air means 21% oxygen. In enclosed or semi-enclosed spaces — shipping containers, tank interiors, pipe galleries, ceiling voids — oxygen can be depleted by rust (oxidation of steel consumes oxygen), biological processes, or displacement by inert gases. Test before entry. Test before hot work. Test continuously during the operation.
How Fires Spread: The Four Mechanisms of Heat Transfer
Understanding the Fire Triangle tells you how fires start. Understanding heat transfer tells you how they grow, spread, and kill. Every fire begins as a small ignition — the threat comes from spread. Fire spreads through four distinct mechanisms, and in real incidents, multiple mechanisms operate simultaneously.
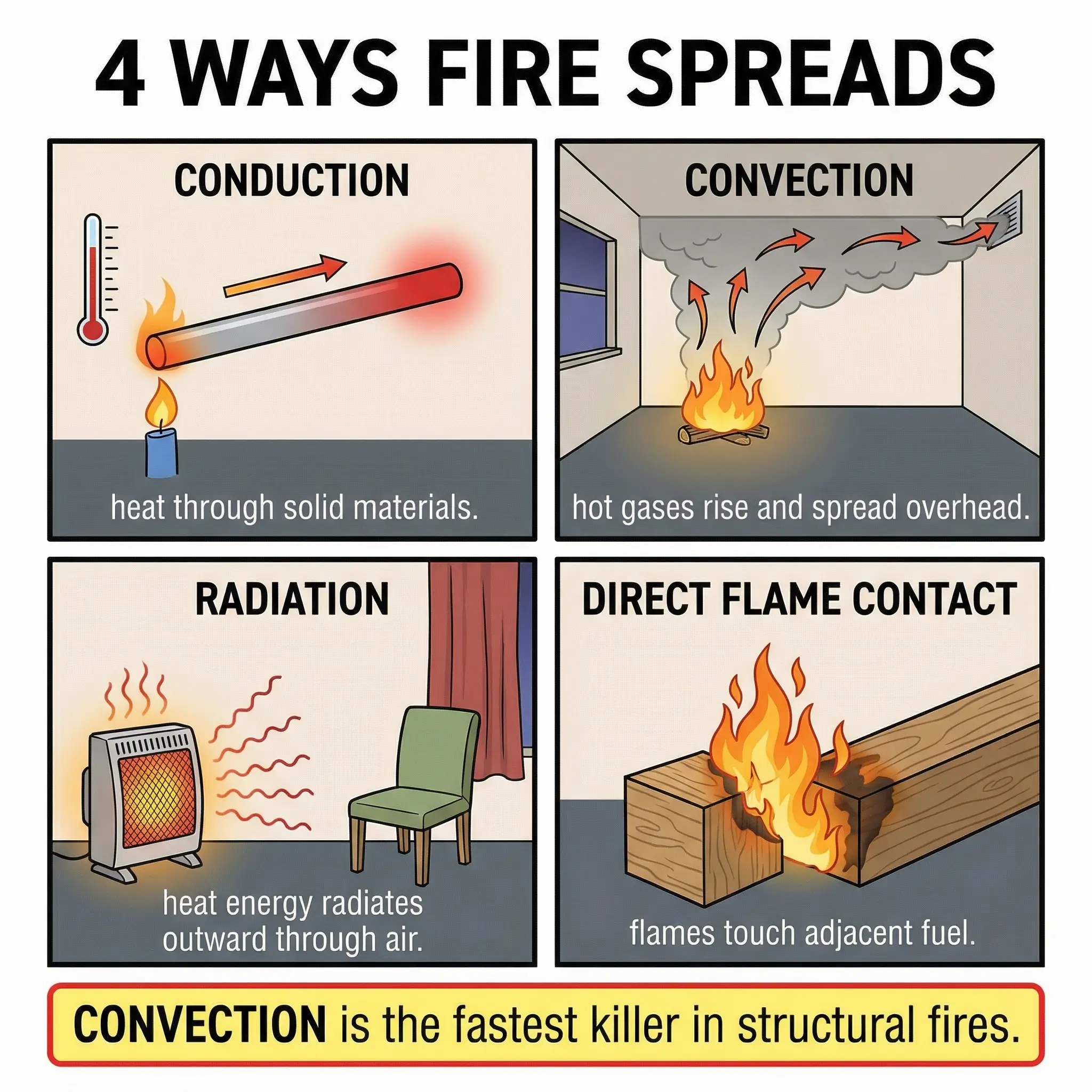
Conduction — Heat Transfer Through Solid Materials
Conduction is heat moving through a solid material from a hot area to a cooler area. Metals are excellent conductors. This is why fires can start on the opposite side of a steel wall from a heat source — the steel conducts the thermal energy directly through the plate.
Field scenarios where conduction causes fire spread include:
- Hot work on steel structures: Welding or grinding on one side of a bulkhead, plate, or pipe while combustible materials contact the other side. The steel surface can reach ignition temperatures meters away from the actual work point.
- Overheating bearings and shafts: A seized bearing heats the shaft, which conducts heat to adjacent packing, seals, or lubricant reservoirs.
- Heated process piping: Steam lines, hot oil circuits, and exhaust ducting can conduct enough heat to ignite insulation, lagging, or adjacent stored materials.
Convection — Heat Transfer Through Hot Gases and Air Movement
Convection is the movement of heat through gases — primarily hot smoke and combustion products rising and spreading. This is the dominant fire spread mechanism in buildings and enclosed structures, and it is the reason most fire fatalities are caused by smoke inhalation rather than burns.
Hot gases rise. In a room fire, a superheated gas layer builds at ceiling level and radiates heat downward onto everything below. When that gas layer reaches approximately 500–600°C, everything combustible in the room ignites simultaneously — a phenomenon called flashover. Flashover is unsurvivable for any unprotected person in the space.
Key convection spread patterns I have observed during fire investigations include:
- Vertical spread through shafts and stairwells: Hot gases funnel upward through elevator shafts, pipe chases, cable trays, and stairwells — spreading fire to floors above the origin faster than horizontal spread on the fire floor.
- Horizontal spread through ceiling voids and ductwork: Concealed spaces above drop ceilings and inside ventilation systems allow fire to travel laterally without visible flame until it breaks through at a remote location.
- Stack effect in tall structures: In high-rise buildings and industrial columns, the chimney effect accelerates vertical convection, pulling fresh air in at the base and pushing hot gases upward at high velocity.
Radiation — Heat Transfer Through Electromagnetic Energy
Radiation is heat energy transmitted through space as electromagnetic waves — similar to how you feel the sun’s heat on your skin. A fire radiates heat in all directions, and any combustible material within the radiation zone can reach its ignition temperature without any physical contact with the flame.
Radiation is the primary mechanism by which fire spreads between separate structures, between storage areas, and across open spaces:
- Tank farm fires: The radiant heat from a single burning storage tank can ignite adjacent tanks if fireproofing, spacing, or water curtain cooling is insufficient. API 2510 and NFPA 30 prescribe minimum spacing distances specifically to manage radiant heat exposure.
- Wildland-urban interface fires: Radiant heat from wildfire fronts ignites structures hundreds of meters ahead of the advancing flame front.
- Warehouse fires: Stacked combustible storage radiates enough heat to ignite goods across an aisle — which is why rack sprinkler systems and aisle spacing are critical in high-piled storage occupancies.
Direct Flame Contact — The Most Obvious Mechanism
Direct flame contact is simply fire spreading by physically touching adjacent combustible materials. While it is the most intuitive form of fire spread, it is also the easiest to control through separation and housekeeping.
Controlling direct flame contact comes down to eliminating fuel continuity — keeping combustible materials physically separated from ignition zones. This is the field principle behind fire breaks, clear zones around hot work, and separation distances in flammable material storage layouts.
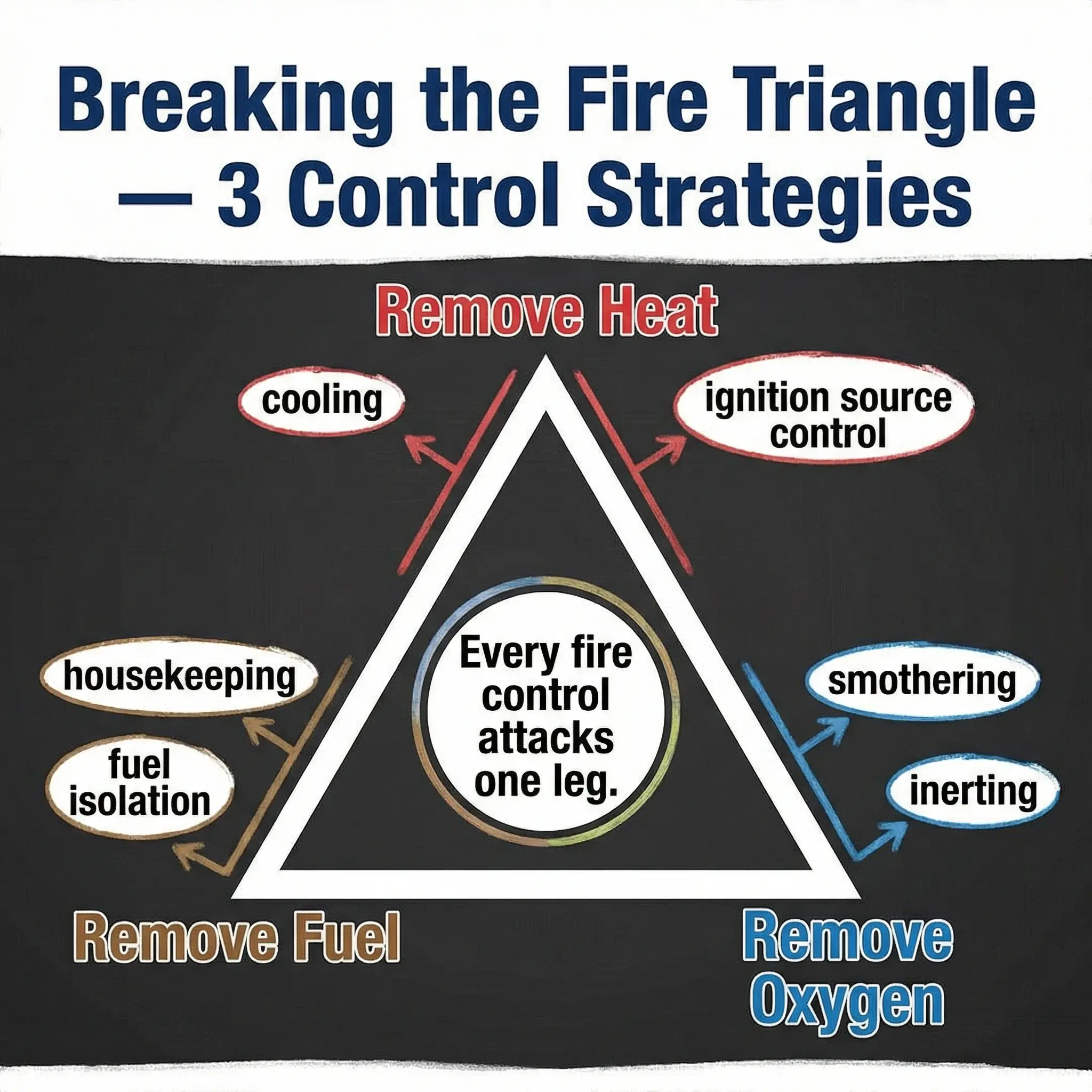
Breaking the Fire Triangle: Practical Prevention and Extinguishment Strategies
Every fire prevention measure and every suppression method works by removing one or more elements of the Fire Triangle. Once you internalize this, fire safety stops being a list of disconnected rules and becomes a coherent system.
Removing Heat — Cooling and Ignition Control
The most common way to fight an active fire is to remove heat. The most effective way to prevent a fire is to control ignition sources before they ever encounter fuel. These strategies target the heat leg of the triangle:
- Water-based suppression: Water absorbs heat energy as it converts to steam (latent heat of vaporization). Sprinkler systems, fire hose streams, and water mist systems all work by cooling the burning material below its ignition temperature. Effective on Class A fires. Never use on Class B liquid fires (water spreads burning liquids), Class C electrical fires (conductivity risk), or Class D metal fires (violent reaction).
- Hot work permit controls: Pre-job fire risk assessments, designated fire watch personnel, removal of combustibles within a minimum 11-meter radius (OSHA 1910.252), post-work fire watch for 30–60 minutes after hot work completion.
- Electrical maintenance programs: Thermographic inspections of panels and connections, proper circuit protection, regular insulation resistance testing, and arc flash hazard analysis per NFPA 70E.
- Preventive maintenance on rotating equipment: Vibration monitoring, lubrication programs, bearing temperature alarms — all designed to catch friction heat buildup before ignition.
Removing Fuel — Elimination and Isolation
Controlling the fuel leg means either eliminating combustible materials from the hazard zone or isolating them so they cannot interact with heat sources:
- Housekeeping discipline: Removing waste, oily rags, packing materials, and combustible debris from work areas — especially around hot work zones, electrical rooms, and process equipment.
- Flammable liquid storage standards: Storing flammable liquids in approved safety cabinets (FM/UL listed), limiting quantities in work areas to one day’s supply, using bonding and grounding during dispensing to prevent static buildup.
- Fuel substitution: Replacing flammable solvents with non-flammable or higher flash point alternatives where process requirements allow.
- Fire-resistant construction: Using non-combustible building materials, fire-rated barriers, fire stops in penetrations, and fire-resistant coatings on structural steel.
Removing Oxygen — Smothering and Inerting
Controlling the oxygen leg is typically used in suppression rather than prevention, though inerting has important prevention applications in specific industries:
- Smothering agents: CO₂ extinguishers displace oxygen around the fire. Fire blankets smother small fires by cutting off air supply. Foam systems float a vapor-suppressing blanket over burning liquid surfaces.
- Inerting: Nitrogen or CO₂ purging of tanks, vessels, and pipelines before hot work to reduce oxygen below combustion-supporting levels. Critical in petrochemical, pharmaceutical, and food processing operations.
- Fixed suppression systems: Clean agent systems (FM-200, Novec 1230) in server rooms, control rooms, and high-value equipment spaces work partially by oxygen displacement and partially by interrupting the chemical chain reaction.
| Extinguishing Method | Triangle Element Removed | Best For | Never Use On |
|---|---|---|---|
| Water (spray/stream) | Heat | Class A — solids | Class B liquids, Class C electrical, Class D metals |
| CO₂ extinguisher | Oxygen | Class B liquids, Class C electrical | Class A deep-seated fires (no cooling), outdoor/windy areas |
| Dry chemical (ABC) | Chemical chain reaction + oxygen | Class A, B, C — multipurpose | Class D metals, sensitive electronics |
| Foam (AFFF/AR-AFFF) | Oxygen (surface blanket) + heat | Class B liquid spills and tanks | Class C electrical |
| Dry powder (Class D) | Heat + oxygen | Class D metals (Mg, Ti, Na, K) | All other classes |
Common Workplace Fire Safety Mistakes I See Repeatedly
After a decade of fire-related inspections, investigations, and audits across multiple industries, certain failures show up on site after site. These are the mistakes that turn manageable risks into actual fires:
- Fire watch assigned but not positioned correctly: The fire watch stands next to the welder watching the arc instead of being positioned where sparks are landing — often below, behind, or on the other side of the work piece. OSHA requires fire watch coverage of “all areas where sparks and hot slag may reach.”
- Combustible material “just out of the way” but still in the hazard zone: I routinely find combustibles pushed to the edge of the 11-meter hot work radius instead of removed entirely. A grinding spark does not respect a tape line.
- Expired or inaccessible fire extinguishers: Monthly inspections missed, extinguishers blocked behind stored materials, wrong class extinguisher for the hazard present. I audited a chemical storage building once that had only a water extinguisher — in a room full of flammable solvents.
- Bypassed or isolated fire suppression systems: Sprinkler systems impaired during construction and never restored. Detection systems in “trouble” or “disabled” mode for weeks. Fire dampers wired open for ventilation convenience.
- No post-hot-work fire watch: The permit says 30 minutes post-work fire watch. The crew leaves immediately because the shift ended. Smoldering fires from hot work have ignited hours after the work was completed — this is how some of the worst industrial fires have started.
- Dust accumulation on elevated surfaces: Combustible dust on cable trays, structural steel, ductwork, and above drop ceilings. A primary explosion suspends settled dust; a secondary explosion from the dust cloud is what destroys the facility.
- Flammable vapor in uninspected low points: Vapors from spills, leaks, or open containers settling into pits, trenches, basements, and sumps where no gas detection exists. One spark in a trench filled with heavier-than-air vapor creates a flash fire.
Pro Tip: Before signing any hot work permit, I physically walk the area and look up, look down, and look on the other side of every wall and floor within the hazard radius. If you cannot see where the sparks and heat are going, you do not know whether it is safe. The permit paperwork means nothing if the field reality does not match.
The Fire Triangle in Emergency Response: Reading Fire Behavior in Real Time
Understanding the Fire Triangle is not only a prevention tool — it is an active decision-making framework during emergency response. When you arrive at a fire scene, you are reading the triangle in real time to determine how to intervene.
Experienced fire response coordinators assess three things immediately, and each maps directly to the triangle:
- What is burning (fuel)? This determines the fire class and dictates which extinguishing agent is safe and effective. Misidentifying the fuel — applying water to a metal fire, for example — can cause a violent reaction.
- How much heat is involved? The intensity and phase of the fire tells you whether portable extinguishers are still viable or whether the fire has escalated beyond manual intervention. Radiant heat that forces you back indicates the fire has exceeded first-response capability.
- Is the oxygen supply being fed or restricted? A fire in an enclosed space may be oxygen-limited and burning slowly — but opening a door or breaking a window introduces fresh air and can cause rapid fire growth or backdraft. This is why forced entry and ventilation decisions during firefighting are life-and-death tactical choices.
During a fire response drill at an offshore platform, I watched a team attempt to fight a simulated engine room fire by opening the compartment door first — flooding the space with oxygen. In a real scenario, that action could trigger backdraft. The correct response for enclosed compartment fires is boundary cooling (removing heat through the boundary) and fixed suppression (removing oxygen with CO₂ or clean agent) — not opening the door and attacking with a hose.
Understanding fire behavior through the lens of the triangle also helps you anticipate what the fire will do next. If you see fire spreading vertically through a cable tray, that is convection driving heat upward into accumulated combustible insulation. If you feel intense heat 20 meters from a visible flame, radiation is the dominant spread mechanism. Each observation points to specific tactical responses — and all of them trace back to which element of the triangle you need to attack.

The Fire Tetrahedron: The Fourth Element
While the Fire Triangle is the practical working model for field safety, modern fire science recognizes a fourth element — the chemical chain reaction — that sustains combustion once it has started. This expanded model is called the Fire Tetrahedron.
The chemical chain reaction involves free radicals produced during combustion that sustain and accelerate the burning process. This is relevant for field practitioners because certain extinguishing agents — dry chemical powder (monoammonium phosphate) and clean agents (FM-200, Novec 1230) — work specifically by interrupting this chain reaction rather than by removing heat, fuel, or oxygen. Understanding this fourth element explains why dry chemical extinguishers are effective on fires where water or CO₂ alone would struggle.
For day-to-day fire prevention planning, hot work control, and hazard identification, the three-element Fire Triangle remains your primary tool. The chemical chain reaction becomes relevant when you are selecting suppression systems, designing fire protection for specific occupancies, or investigating why a particular extinguishing method failed.
Fire Triangle Application in Permit-to-Work Systems
Every effective hot work permit system is, at its core, a structured method of verifying that the Fire Triangle has been broken before work begins. When I review or design permit-to-work systems, I map every permit requirement to the specific triangle element it controls.
The following table shows how standard hot work permit requirements directly target elements of the Fire Triangle:
| Permit Requirement | Triangle Element Controlled | Field Application |
|---|---|---|
| Gas testing for flammable atmospheres | Fuel (vapor presence) | Confirm LEL < 10% before ignition source introduced |
| Removal of combustibles within 11m radius | Fuel (solid/liquid) | Physical removal or fire-resistant covering |
| Fire-resistant blankets/shields | Fuel (protection) | Shielding combustibles that cannot be removed |
| Fire watch with charged extinguisher | Heat (rapid response) | Immediate suppression if ignition occurs |
| Verification of sprinkler system operability | Heat (automatic suppression) | Confirm system is live and unimpaired |
| Post-work fire watch (30–60 minutes) | Heat (delayed ignition) | Monitor for smoldering ignition after heat source removed |
| Inerting of tanks/vessels before hot work | Oxygen (atmosphere control) | Reduce O₂ below combustion threshold |

Conclusion
The Fire Triangle is not a poster on a training room wall. It is a real-time decision-making tool that every worker, supervisor, and safety professional should use every day — during inspections, before hot work, when storing materials, when reviewing hazards, and when responding to emergencies. Heat, fuel, and oxygen are present in every workplace. Your job is to make sure they never meet uncontrolled.
Every fire I have investigated — from small smoldering fires caught by alert workers to major incidents that shut down entire operations — traces back to the same failure: the three elements of the triangle converged because someone did not identify one of them, did not control one of them, or did not verify that controls were still in place. Fire prevention is not about perfection. It is about discipline. Check the triangle. Walk the area. Remove the fuel. Control the heat. Verify the atmosphere. Do it every time, not just when someone is watching.
The best fire response is the one that never happens. And the simplest, most proven framework for making sure it does not happen is the same one fire professionals have relied on for generations — break the triangle, and fire cannot exist. That responsibility does not sit with the safety department alone. It belongs to everyone who steps onto a site where heat, fuel, and oxygen are present. Which means it belongs to everyone.
Note: Conducting fire risk assessment training ensures that individuals are equipped with the knowledge and skills to identify hazards and apply proper fire control measures. Fire prevention is not just about extinguishing flames but also about minimizing risks and ensuring a safer environment in homes, workplaces, and industrial settings. By staying informed and prepared, we can reduce fire risks, protect lives, and safeguard property.

